Aim of study: Curcuma caesia (Family: Zingiberaceae) is being used in traditional medicine for the treatment of severe bronchitis and asthma. The aim of study was to evaluate antiasthmatic activity of various extracts of curcuma caesia rhizomes to validate its traditional use. Materials and methods: In the present study petroleum ether, ethanol and water extract of Curcuma caesia rhizomes at the doses of 25-100 mg/kg i. p. was evaluated for antiasthmatic activity using clonidine induced mast cells degranulation in mice. Results: Treatment group of CCEE (Curcuma caesia rhizomes ethanol extract) exhibited significant mast cell stabilizing potential at all three doses (25, 50, 100 mg/kg i.p) 27.2±1.854, 23.4±1.166, 17.6±1.452 respectively as compared to control group. Percent protection offered by CCEE at all three doses was notably 75.13%, 76.07%, 81.70% and was of statistical significance as compared to CCPEE and CCWE. Conclusion: It can be concluded that the ethanol extract of Curcuma caesia (CCEE) may be useful in management of asthma.
Keywords:Curcuma caesia, asthma, mast cell degranulation.
Asthma is a chronic inflammatory isorder of airway. Asthma affects about 300 million people worldwide and it has been estimated that a further 100 million will be affected by 2025. Asthma is common global health problem among individual of all ages are offered by chronic airway disorder. It defines as a chronic reactive airway disorder that produces episodic reversible airway obstruction via bronchospasm, increased mucous secretions and mucosal edema. Asthma is a chronic inflammatory disorder of the airways.[1]
1. WHO estimates show that 235 million people currently suffer from asthma.
2. Asthma deaths will increase in the next 10 years if urgent action is not taken. Asthma cannot be cured, but proper diagnosis, treatment and patient education can result in good asthma control and management.
3. Asthma occurs in all countries regardless of level of development. Over 80% of asthma deaths occur in low and lower-middle income countries. For effective control, it is essential to make medications affordable and available, especially for low-income families.
4. Asthma is a chronic disease characterized by recurrent attacks of breathlessness and wheezing, which vary in severity and frequency from person to person.
5. Symptoms may occur several times in a day or week in affected individuals. For some people the symptoms become worse during physical activity or at night. Failure to recognize and avoid triggers that lead to a tightened airway can be life threatening and may result in an asthma attack, respiratory distress and even death.
6. Through appropriate treatment such as using inhaled corticosteroids to ease bronchial inflammation, the number of asthma-related deaths can be reduced.
7. Asthma is the most common chronic disease among children. But it can be controlled through different prevention and treatment plans according to individual symptoms.
8. The strongest risk factors for developing asthma are exposure to indoor allergens such as house dust mites in bedding, carpets and stuffed furniture; pollution and pet dander; outdoor allergens such as pollens and moulds; tobacco smoke and chemical irritants in the workplace.
9. Asthma triggers can include cold air, extreme emotional arousal such as anger or fear, and physical exercise.
10. Asthma is often under-diagnosed and undertreated, creating a substantial burden to individuals and families and possibly restricting individuals’ activities for a lifetime.[2]
Curcuma caesia Roxb. is a member of the family Zingiberaceae and popularly known as Kali haldi. In India it is found in West Bengal, Madhya Pradesh, Orissa, Chhattisgarh, and Uttar Pradesh states. It nourishes well in moist deciduous forest areas. Rhizomes of the plant are used for sprains and bruises and also employed in the preparation of cosmetics.The effective use of Curcuma longa Linn. well known since a long time; it is laxative, anthelmintic, and vulnerary, besides this it is used in blood disorders, leukoderma, scabies, small-pox, and sprains. Curcuma amada Roxb. is useful in bronchitis, asthma, sprains, skin diseases, and in ammation caused due to injuries. The genus Curcuma is a well-known spice of India. It is also called Haldi and more than 200 species and subspecies of it is found all across the world. One of which is Curcuma caesia Family: Zingiberaceae. It is also known as “Kali Haldi.” It is an erect rhizomatous herb with large leaves. Fresh rhizomes are aromatic with intense camphoraceous odour and are applied externally to sprain and bruises. Black Turmeric (Curcuma caesia) is native to North-East and Central India. It is also sparsely found in Papi hills of East Godavari, the root hills of the Himalayas and North Hill forest of Sikkim. The rhizomes of Black Turmeric have a high economic importance owing to its putative medicinal properties.[3,4] Rhizome large, 5-6 × 9- 10 cm, (Figure 1) blue in the centre, verging towards grey, the blue colour is highly variable, depending upon the nature of the soil and age of the rhizome, strongly aromatic; sessile tubers branched, condensed; roots fleshy; root tubers many, ovate oblong, pale, watery pearl colour. Plants large, 70-100 cm tall, pseudo stem 30-35 cm tall, sheaths green. Leaves distichous, (Figure 2) 79-100 cm; petiole as long as lamina; lamina 30-40 × 10-12 cm, oblong lanceolate, tip acute, base acuminate, glabrous, purple or reddish brown patch along the sides on the distal half of the mid rib on upper side only, fading at maturity, groove of the midrib green. Inflorescence lateral, 25-30 cm long, peduncle 12-18 cm; spike 12-15 × 5 cm; coma bracts large, pink to violet, lower ones streaked green. Fertile bracts 18- 20, 4.5-5 × 4.4-5 cm, lower half used, tip rounded, green with pink tip, each bract subtends a cincinnus of 8-10 flowers.[5,6] The object of the present study was to evaluate antiasthmatic activity of various extracts of curcuma caesia rhizomes by using clonidine induced mast cell degranulation in mice.
Plant material
Rhizomes of Curcuma caesia were collected in Feb 2012, from Botanical Garden (Zinger Villa) Calicut University, Calicut (Kerala, India), and the plant was authenticated by Professor Dr. M. Sabu, Head, Department of Botany, Calicut University, Calicut (Kerala, India). A voucher specimen (PAR 14) was deposited in the herbarium for further use.
Extraction
Dried and coarsely powder of Curcuma caesia rhizomes (100 g) was defatted with petroleum ether and the marc remaining was extracted successively by petroleum ether, ethanol and water in different Soxhlet extractor. Solvent was evaporated in rotary evaporator under reduced pressure.
Animals
Swiss albino mice of either sex weighing (25-30 g) were housed under standard laboratory condition of temperature (25 ± 2°C) and 12/12 h light/dark cycle. The animals had free access to food and water. The Animal Ethical Committee of the Institute approved all the protocols of the study.
Drugs and Chemicals
5% polyethylene glycol (PEG-400), saline solution, buffer medium, Sodium chromoglycate, RPMI-1640 were purchased from Himedia, India. Clonidine (Alembic), petroleum ether (60-80°C), ethanol (95%). All Chemical and reagents were of analytical grade.
Acute toxicity study
Acute oral toxicity study was performed as per OECD 423 guideline. Extract was administered up to the maximum dose of 2000 mg/kg and animals were observed for mortality.[7]
Clonidine induced mast cell degranulation in mice
Mice were divided into five groups, five animals in each group. A three-day drug treatment schedule was followed. Group-I serves as control receives vehicle only (5 % PEG400, 1ml / kg, i.p.). Group-II receives standard drug disodium cromoglycate (DSCG, 200µg/kg, i.p.). Group-III, IV and V was treated with test extract 25, 50 and 100 mg/kg, i.p. respectively. On day fourth, each animal was injected with 4 ml/kg, 0.9 % saline solution, into peritoneal cavity. By gentle massage, peritoneal fluid was collected after 5 mins and transferred into siliconised test tubes containing 7-10 ml RPMI-1640 buffer medium (pH 7.2-7.4). This solution was then centrifused at 400-500 RPM. Pellet of mast cell was washed with same buffer medium twice by centrifugation, discarding supernatant. These cells were challenged with clonidine (50 g), incubated at 37oC in a water bath for 10 mins. Followed by staining with 1% toluidine blue and observed under microscope (10X). Total 100 cells were counted from different visual area. Percent protection against degranulation was calculated.[8,9]
Statistical analysis
The data were presented as mean ± Standard error mean. The statistical significance between the groups has been tested by analysis of variance followed by Dunnett’s test. A P < 0.05 were considered as significant.



Acute toxicity test
The LD50 value of Curcuma caesia rhizome extracts when given intraperitoneally and tested in albino mice was found to be more than 2000 mg/kg body weight.
Clonidine induced mast cell degranulation
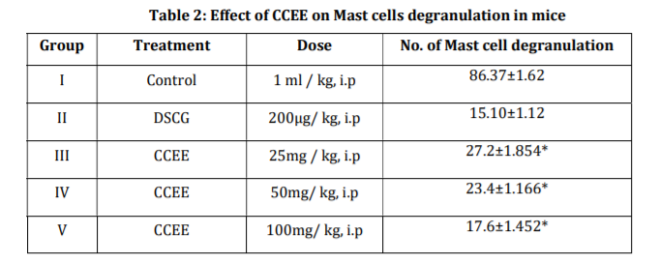
Treatment group of CCEE (Curcuma caesia rhizomes ethanol extract) exhibited significant mast cell stabilizing potential at all three doses (25, 50, 100 mg/kg i.p) 27.2±1.854, 23.4±1.166, 17.6±1.452 respectively (Table 2 and figure 2) as compared to control group. Percent protection offered by CCEE at all three doses was notably 75.13%, 76.07%, 81.70% and was of statistical significance as compared to Curcuma caesia Petroleum ether extract (CCPEE) and Curcuma caesia water extract (CCWE). CCPEE at 100 mg/kg shows 63.40±1.496 mast cells degranulation (Table 1 and Figure 1) while CCWE at 100 mg/kg shows 65.8±0.663 mast cells degranulation (Table 3 and Figure 3) which was not-significant.
Mast cells are the principal store of histamine and other many chemical mediators responsible for the pathogenesis of asthma. Mast cells are necessary for the development of allergic reactions, through cross liking of their surface receptor for IgE, leading to degranulation and the release of vasoactive, pro-inflammatory and nociceptive mediators. Released mediators from mast cells, each having more than one potent effect on airway inflammation.[10] Upon histological examination, decreased numbers of granulated mast cells have been observed in the airways of patients who died from acute asthma attacks; indicate that mast cell degranulation is a contributing factor in the progression of disease.[11] There is an increased presence of mast cell in the airway smooth muscle in-patient with asthma, because the attraction caused by the secretion of stem cell factor by the muscle. Stem cell factor is chemo attractant for mast cell and is responsible for regulating their growth, function, and survival. Mast cells degranulation is important in the initiation of immediate responses following exposure to allergens.[12] Almost 3-5 times increase in mast cells inpatient of asthma. Once binding of allergen to cell-bound IgE occurs, mediators such as histamine, eosinophils, neutrophils, chemotactic factor LTC4, LTD4, and LTE4, PG, and PAF etc. are released from mast cells. Mast cell degranulation is believed to be an integral cause of exercise-induced bronchospasm following either drying or cooling of the airways.[13] It is known that clonidine releases histamine from mast cells. Clonidine, a α2 adrenoreceptor agonist, induces mast cell degranulation same as of compound 48/80, causes the expulsion of granules without any damage to cell wall in mice. The underlining principle is number of mast cells collected from the peritoneal fluid of mice that degranulate when challenged with clonidine and the percent protection offered by the test extracts compared with standard drug disodium cromoglycate (DSCG) observed under microscope (45 X).[9] in our previous study it was observed that EECC at doses of 100-150 mg/kg significantly decreases milk induced eosinophils and leucocutosis count in dose dependant manner.[5,6] present study revealed that ethanolic extract of Curcuma caesia shows significant role in mast cell protection against clonidine induced degranulation, while petroleum ether and water extract of Curcuma caesia rhizomes was nonsignificant.
Ethaniolic extract of Curcuma caesia rhizomes offers mast cell protection against degranulation as compared to petroleum ether and water extracts. It leads to conclusion that CCEE may be useful in the management of asthma.
References
- 1.Kabesch M, Schedel M, Carr D, et al. IL-4/IL-13 pathway genetics strongly infl uence serum IgE levels and childhood asthma. J Allergy Clin Immunol, 117 (2): 269 – 274, 2006. [Google Scholar] [PubMed]
- 2.World Health Organization. Global surveillance, prevention and control of chronic respiratory diseases: a comprehensive approach, 2007. [Google Scholar] [PubMed]
- 3.Aslam R. Pathan, Gautam Vadnere, M.Sabu. Curcuma caesia almost untouched drug: an untouched Ethnopharmacological review. Inveni rapid: Planta Activa, 4: 1-4, 2003. [Google Scholar] [PubMed]
- 4.M Sabu. Zingiberaceae and Costaceae of South India. Kerala: Indian Association for Angiosperm Taxonomy, Department of Botany, Calicut University, Kerala, India; p.132-138, 2006. [Google Scholar] [PubMed]
- 5.Aslam Ramjan Pathan. Pharmacological Investigation of Curcuma caesia in Management of Asthma. Inventi Rapid: Ethnopharmacology, 2014(4):1-4, 2014. [Google Scholar] [PubMed]
- 6.Aslam Ramjan Pathan. Ethnopharmacological Evaluation of Curcuma caesia in Management of Asthma. Inventi Rapid: Ethnopharmacology, 2014(4):1-4, 2014. [Google Scholar] [PubMed]
- 7.Organization of Economic Co-operation and Development (OECD). The OECD Guidelines for Testing of Chemical: 423 Acute Oral Toxicity, France, 2001. [Google Scholar] [PubMed]
- 8.Gupta P, Srimal R. Passive cutaneous anaphylactic inhibitory and mast cell stabilizing activity of Coleonol and its derivatives. Indian J of Pharmacol 1994; 26: 150 – 152. [Google Scholar] [PubMed]
- 9.Lakdawala AD, Dadkar NK, Dohadwalla AN. Action of clonidine on the mast cells of rats. J. Pharm. Pharmacol. 1980; 32: 790-791. [Google Scholar] [PubMed]
- 10.Theoharis C, David E. Critical role of mast cells in inflammatory disease and the effect of acute stress. Neuropharmacology 2004; 146: 1-12. [Google Scholar] [PubMed]
- 11.Serafin WE. Drugs used in the treatment of Asthma. In: Goodman Gillman A. (Eds.) the Pharmacological Basis of Therapeutics. 9th edn. New York: McGraw Hill; 1996: 661. [Google Scholar] [PubMed]
- 12.Bethesda N, National Asthma Education and Prevention Program Expert Panel Report: 2, Guidelines for the Diagnosis and Management of Asthma. NTH Publication No.1997; 97:4051. [Google Scholar] [PubMed]
- 13.McFadden E, Elsanadi N, Dixon L. Protocol therapy for acute asthma: Therapeutic benefits and cost savings. Am. J. Med. 1994; 99: 651-661. [Google Scholar] [PubMed]

